Product
We are pioneering the future of operating room air safety through our hands-free smoke and bioaerosol capture technology. Our Flamingo Funnel™ family of devices outperforms traditional smoke evacuation pens—removing nearly twice as much smoke1—while offering a hands-free, non-obstructive footprint optimized for modern surgical workflows.

Three available sizes
Our hands-free smoke and bioaerosol capture technology is available in three sizes to accommodate different surgical procedures. The flexible design of the Flamingo Funnel allows the device to conform to the patient’s unique anatomy, keeping the device – and smoke – out of sight.

Part #: SQ20012-01 | Plenum Size: 9.0″ wide
Suggested Clinical Use:
Spine, Thoracic, Laparotomy, Hip (lateral or anterior), Breast lumpectomy with axillary lymph node sampling.

Part #: SQ20012-02 | Plenum Size: 6.25″ wide
Suggested Clinical Use:
Shoulder, Hip, Ventral hernia, GYN including C-Section, Lumbar Spine decompression fusion (Note: Turn off suction before uterus is entered).
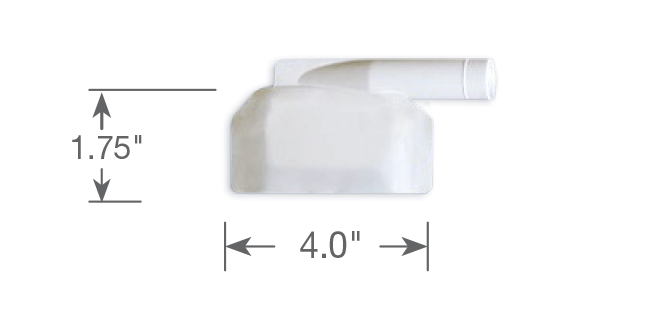
Part #: SQ20012-03 | Plenum Size: 4.0″ wide
Suggested Clinical Use:
Head and neck surgeries, Inguinal and umbilical hernias, Lumps and bumps, Shorter incisions in outpatient surgeries, Endoscopic spine cases, Breast mass excision, Perineal cases (anal, vaginal), Lumbar Spine decompression fusion.

Allows for two Flamingo Funnels to be attached to a single smoke evacuator. All ports are 22 mm O.D.
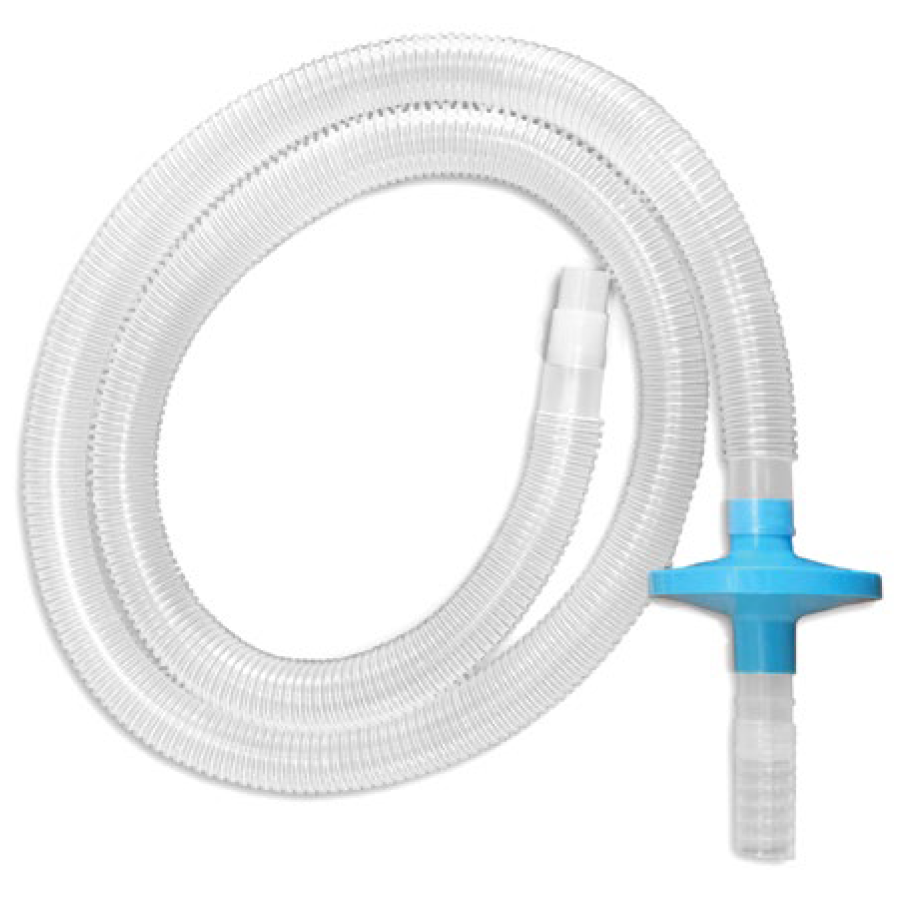
72” of tubing connects to the Flamingo Funnel to extend the range for placement of the smoke evacuator.
FAQ
There are several health hazards that have been identified with surgical smoke:
- Surgical smoke plumes contain toxic gases, bioaerosols, and carcinogens
- OR staff are exposed to the equivalent of smoking 27 to 30 cigarettes daily
- Surgical teams can develop respiratory issues, infections, and other long-term health problems
Additionally, smoke capture is now mandated in 20+ states. Click here for the most up to date guidance.
A smoke evacuation system has 2 parts – the smoke evacuator (a vacuum pump like a Neptune) and the smoke capture device (a Flamingo Funnel or ESU pencil). The Flamingo Funnel can be used with any commercially available smoke evacuator.
Open surgical procedures require a smoke capture device like the Flamingo Funnel or an ESU pencil with integrated smoke capture in order to remove smoke from the operating room. Laparoscopic procedures require a different type of smoke capture device that is unique to this type of procedure and different than the Flamingo Funnel.
The Flamingo Funnel is a hands-free smoke capture device that does not obstruct the surgeon’s view. In addition, it has been shown to capture up to two times more surgical smoke than ESU pencils1 with integrated smoke capture and to be effective at removing infection-causing bioaerosols2,3.
Yes, the Flamingo Funnel can be used with any type of cautery device in open surgical procedures in order to capture the surgical smoke.
We recommend setting the smoke evacuator at 100% continuous vacuum power level prior to starting the procedure to ensure you get the full smoke capture benefit of the Flamingo Funnel. You can adjust power levels downward if less smoke capture is required.
Yes, the Flamingo Funnel works with all brands/models of smoke evacuators. The connector on the end of the Flamingo Funnel is designed to fit the standard 7/8” size vacuum port size found on smoke evacuators.
Yes, two Flamingo Funnels can be used at the same time when there will be more than one incision and the Flamingo Funnel cannot be easily moved during the procedure. The Flamingo Funnel T-Connector (P/N SQNS10011-01) can be used to connect two medium or small Flamingo Funnels to a single smoke evacuator.
We recommend using the Flamingo Funnel size that is at least as wide as the length of the planned incision. For example, if the incision will be approximately 4” long, then we recommend using the medium size of the Flamingo Funnel (4.25” wide).
The leading edge of the Flamingo Funnel (with the exposed foam) should be parallel to and within .5-1.5” of the planned incision. The closer the device is to the incision, the more effective it will be at capturing surgical smoke.
Once the leading edge of the device is more than 1.5” away from the incision, the smoke capture effectiveness starts to diminish significantly.
The Flamingo Funnel works well in any open surgical procedure where the leading edge of the device can be within .5-1.5” of an incision.
For procedures where the Flamingo Funnel cannot be within .5-1.5” of the entire incision, consider moving the Flamingo Funnel during the procedure to stay with .5-1.5” of the incision or placing a second Flamingo Funnel on the patient.
The Flamingo Funnel can be placed directly on the skin or on top of a drape or Ioban drape. The device can also be partially on top of an Ioban drape and partially on top of the skin if necessary to get the leading edge of the device within .5-1.5” of the incision (i.e., when the edge of the Ioban drape if more than 1.5” away from the incision). Note that the Ioban drape may tear if you try to remove the device. If you plan to move the device during a procedure we suggest placement on the skin or outer drape.
Any type of drape (Ioban, loose surgical drape, etc.) can be placed on top of the device as long as the leading edge of the Flamingo Funnel is not blocked.
We do not recommend placing the device on top of a loose outer surgical drape since the weight of the device might move the drape and the device away from the incision during the procedure and/or the drape may bunch up and block the leading edge of the device.
Ultimately, there is a lot of flexibility in terms of how to drape around the device as long as the leading edge is not blocked and is within .5-1.5” of the incision.
Using the Flamingo Funnel in procedures that require retractors should not be an issue in most situations. Even if the retractors block a small portion of the leading edge of the Flamingo Funnel (where the foam is exposed) or if a portion of the Flamingo Funnel is deformed/squashed by retractors, the device will still effectively capture surgical smoke.
Yes, the device is intended to be flexible so that it can conform to patient anatomy. It can easily bend without any damage to the device.
Yes, the Flamingo Funnel can be moved during the procedure if necessary. If the device has been put directly on the patient’s skin or on a drape, the sticky backing can typically be repositioned at least once during the procedure. If the sticky backing doesn’t secure it well, consider clamping the wings of the device to the drape. If the Flamingo Funnel has been applied on top of an Ioban drape, then trying to separate it from the drape and move it during the procedure is likely to lift and/or damage the drape.
You can also use two Flamingo Funnels in different places as needed. They can be connected with a T-Connector.
The adhesive used on the Flamingo Funnel is safe to use on patient skin. Our customers have not experienced any issues with skin damage during removal of the device.
The larger Flamingo Funnel tubing is specifically designed to allow for the high flow rates that can be achieved with the device. This is part of what makes the smoke capture so effective with the device.
Small amounts of blood and fluids will not impact the performance of the Flamingo Funnel.
We have a variety of learning resources available on our website at: https://flamingofunnel.co/instructions/
In addition, we are available for in person support if you contact us at: [email protected].
1 Schultz L. An Analysis of Surgical Smoke Plume Components, Capture and Evacuation. AORN Journal. Feb 2014, Vol 99; No 2.
2 Liu et al., The Utility of Local Smoke Evacuation in reducing Surgical Smoke Exposure in Spine Surgery: A Prospective self-controlled study. The Spine Journal. 20 (2020), 166-173.
3 Krueger, S., Disegna, S., & DiPaola, C. (2018). The effect of a surgical smoke evacuation system on surgical site infections of the spine. Clinical Microbiology and Infectious Diseases, 3(1), 1–5.
