Capturing and evacuating surgical smoke from the operating room remains a critical issue. Surgical smoke not only compromises air quality for clinical staff but also introduces safety, visibility, and compliance concerns.
Many states have enacted regulations — often aligned with AORN’s recommendations — requiring healthcare facilities to implement effective smoke evacuation systems in operating rooms, making smoke capture a compliance and safety necessity.
Surgical smoke contains hazardous by-products such as toxic chemicals, particulates, and bioaerosols that can irritate the respiratory tract, reduce visibility, and contribute to long-term health risks for surgical staff. Additionally, bioaerosols can be harmful to surgical staff and patients, increasing surgical site infections.
While standard smoke capture solutions like ESU-integrated smoke capture pens are commonly used to remove smoke from the surgical field, they are not without drawbacks: these tools can reduce surgeon dexterity, obstruct the field of view, and may not always generate sufficient suction to reliably capture smoke at the source.

Introducing
Flamingo Funnel™
Clean air. Clear view.
- Hands-free
- Unobstructed view
- Flexible design
- Up to 2x the smoke capture1
-
Shown in studies to remove infection-causing bioaerosols2,3

The Flamingo Funnel is available in three sizes to fit different surgical procedures and flexes to fit patients’ unique anatomy.
Hands-free Flamingo Funnel™
vs. the competitionFlamingo Funnel
ESU Pencil
Average Smoke Capture Efficiency (%)*
99.5%
51.4%
Hands-free
No Vision Obstruction
No Hand Fatigue
Reduces bio-aerosols
Disposable
Works with alternative cautery sources, i.e., bipolar, laser & plasma

“OR smoke is dangerous to OR staff health. I use the Flamingo Funnel because it has been shown in studies to remove more smoke and bioaerosols than the ESU pencils.”
Dr. Sunny Kim
inspired Spine
Conducting a surgical smoke capture evaluation
As healthcare teams increasingly look to quantify and improve their smoke-management practices, it is imperative to conduct thorough evaluations of smoke capture solutions.
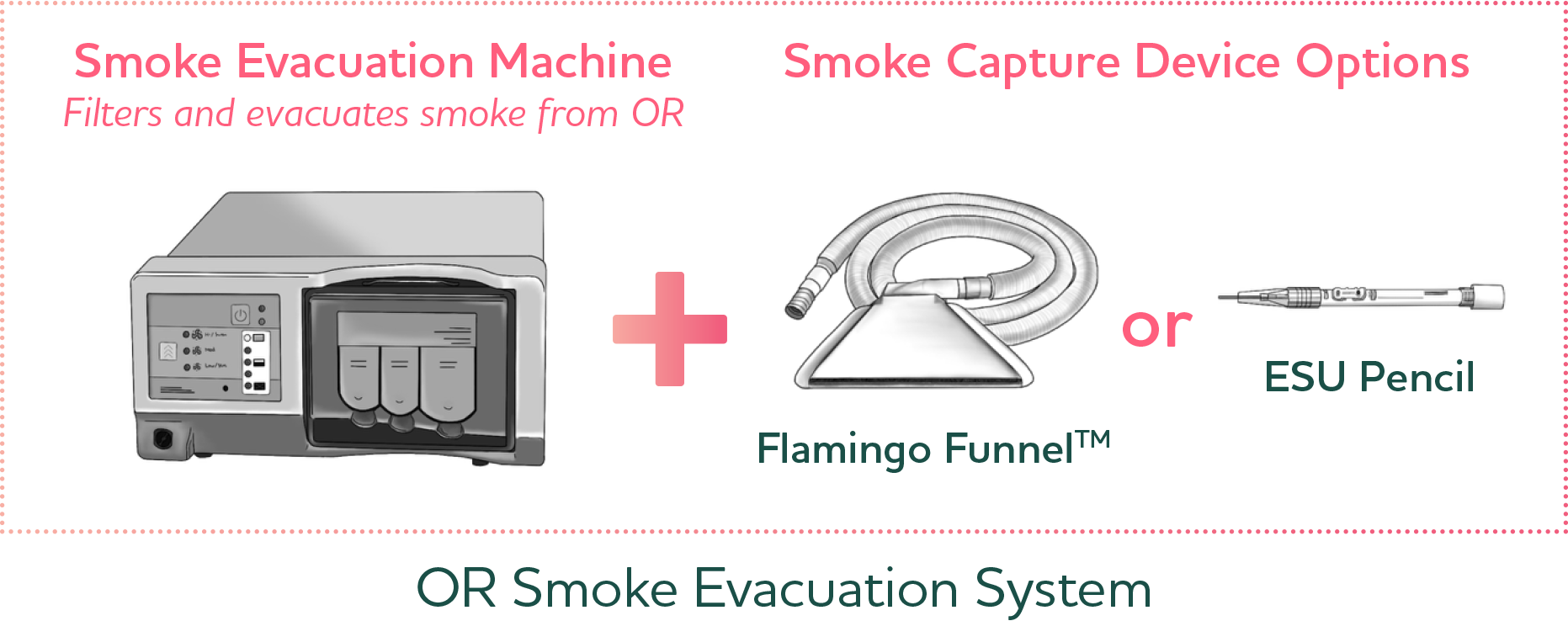
Smoke capture vs. smoke evacuation
A smoke evacuation system has two parts – the evacuator (a vacuum pump) – and the smoke capture device (the Flamingo Funnel or ESU pencil). When it comes to smoke capture, one size does not fit all. It is important to evaluate multiple options to ensure you have the right solutions for various surgical needs.
Considerations for evaluating smoke capture devices
When selecting your smoke capture device for your smoke evacuation system, try multiple types of devices to ensure they meet your various surgical needs and consider:
- Is it a mandate-compliant solution?
For smoke capture compliance guidelines, reference AORN’s Guidelines for Perioperative Practice.
For smoke evacuation compliance guidelines, reference ISO 16571:2024(en): Systems for evacuation of plume generated by medical devices.
- Level of smoke capture required, dictated by the amount of smoke created
- Need for visualization or dexterity
- Ease of use
- Surgeon preference
- Effectiveness of the solution for smoke and bioaerosol capture
Key steps to prepare for your evaluation

Identify interested surgeons and/or surgical techs and designate “super users” to be trained for longer evaluations. Make sure to recruit a cross-section of surgeons from service lines throughout your organization.

Different types of surgery produce different amounts of smoke and may have unique needs. In the same vein, some surgeries require increased fine motor skills where dexterity and visibility are paramount. Some smoke capture devices may address these needs better than others.

Clearly educate the team about the device features, how to use it effectively and its supporting data. Ensuring the surgical team knows how to properly use the device is key to a successful evaluation.

Pre-scheduling time to discuss usage and placement is crucial for success. Ask surgeons to speak to the device representative and/or surgical tech approximately 15 minutes before the procedure begins to discuss product use and placement.
Best practices
Test one type of smoke capture device per day
This prevents cognitive overload for surgical staff, reduces bias, and ensures that feedback reflects true performance rather than comparison fatigue.
Ensure each device gets a meaningful number of uses
Each smoke capture device should be evaluated across multiple cases or shifts so surgical staff can see how it performs under different conditions (simple, complex, long, short procedures).
Rotate devices across comparable days
If testing multiple competing products, evaluate them on separate days with similar case types, staffing, and patient complexity so performance data is comparable.
Use the device consistently throughout each procedure
Ensure the surgical team uses the smoke capture device in the same manner and at the same stages of the procedure each time. This consistency allows evaluators to compare performance across cases reliably and prevents variations in technique from influencing results.
Capture structured feedback immediately after each us
Real-time impressions are more accurate than end-of-day or retrospective recall. Assign someone to collect and record feedback in the OR.
Post-evaluation analysis
Schedule a post-evaluation meeting with the team to review results and discuss next steps, such as risk analysis, surgical needs and preferences that may drive decision making.
Include the Flamingo Funnel in your evaluation
To include the Flamingo Funnel in your smoke capture evaluation, please contact us: [email protected].
Download our surgical smoke capture evaluation tool!
1 Schultz L. An Analysis of Surgical Smoke Plume Components, Capture and Evacuation. AORN Journal. Feb 2014, Vol 99; No 2.
2 Krueger, S., Disegna, S., & DiPaola, C. (2018). The effect of a surgical smoke evacuation system on surgical site infections of the spine. Clinical Microbiology and Infectious Diseases, 3(1), 1–5.
3 Liu et al., The Utility of Local Smoke Evacuation in reducing Surgical Smoke Exposure in Spine Surgery: A Prospective self-controlled study. The Spine Journal. 20 (2020), 166-173.
MKT-SSS-0035
